In the realm of oncology, early detection of cancer remains paramount in improving patient outcomes. Traditionally, cancer diagnosis has relied heavily on invasive procedures such as biopsies and imaging scans. However, recent advancements in medical technology have paved the way for a less invasive approach – blood tests for cancer detection. These tests, often referred to as liquid biopsies, analyze circulating tumor cells (CTCs), circulating tumor DNA (ctDNA), and other biomarkers present in the bloodstream, offering a promising avenue for early cancer detection, monitoring, and personalized treatment strategies.
Understanding Liquid Biopsies:
Liquid biopsies involve the analysis of various components present in the blood, including CTCs, ctDNA, extracellular vesicles, and tumor-derived proteins. These components shed light on the molecular characteristics of tumors, providing valuable insights into their genetic mutations, tumor heterogeneity, and metastatic potential. Unlike traditional tissue biopsies, which may be limited by tumor accessibility and invasiveness, liquid biopsies offer a non-invasive and convenient method for cancer detection and monitoring.
Advantages of Blood Tests for Cancer Detection:
Early Detection: One of the most significant advantages of blood tests is their potential to detect cancer at an early stage when treatment options are more effective and outcomes are favorable. By identifying molecular changes associated with cancer in the bloodstream, these tests can facilitate early intervention and improve patient survival rates.
Minimal Invasiveness: Unlike tissue biopsies, which require surgical procedures to obtain samples, blood tests for cancer detection are minimally invasive and pose fewer risks to patients. This non-invasive approach reduces patient discomfort, eliminates the need for anesthesia, and allows for serial monitoring of disease progression and treatment response.
Real-Time Monitoring: Blood tests for cancer detection enable real-time monitoring of disease progression and treatment response, providing oncologists with valuable insights into tumor dynamics and therapeutic efficacy. By tracking changes in circulating tumor biomarkers over time, clinicians can make informed decisions regarding treatment adjustments and personalized therapeutic strategies.
Personalized Treatment Strategies: Liquid biopsies provide a comprehensive molecular profile of tumors, allowing for the identification of specific genetic mutations, tumor markers, and therapeutic targets. This molecular characterization enables oncologists to tailor treatment regimens to individual patients, maximizing treatment efficacy while minimizing potential side effects.
Accessibility and Convenience: Blood tests for cancer detection offer unparalleled accessibility and convenience, as they can be performed in a clinical setting using routine blood samples. This accessibility makes them particularly well-suited for population-wide screening programs and surveillance strategies aimed at high-risk individuals, such as those with a family history of cancer or genetic predisposition to certain malignancies.
Emerging Technologies and Applications:
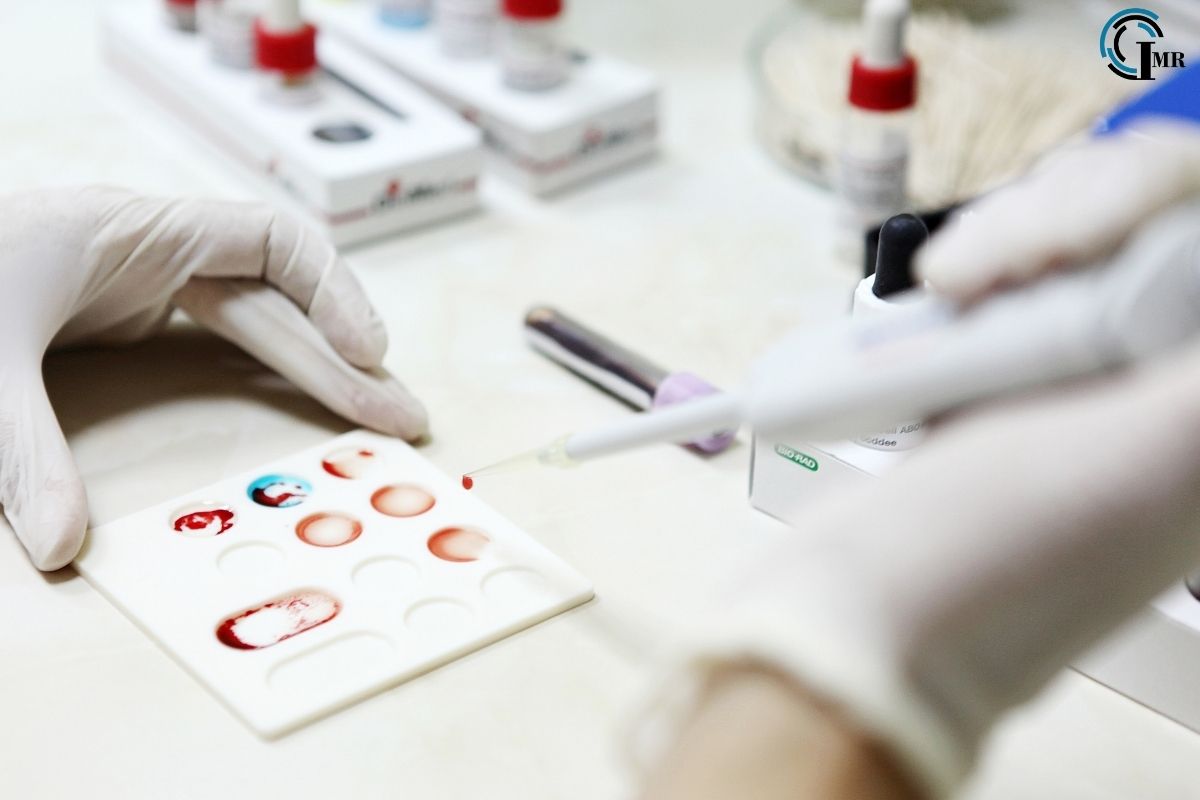
Next-Generation Sequencing (NGS): NGS technologies have revolutionized the field of liquid biopsy by enabling comprehensive genomic profiling of tumors from circulating DNA fragments. These high-throughput sequencing platforms can detect a wide range of genetic alterations, including point mutations, insertions/deletions, copy number variations, and chromosomal rearrangements, with high sensitivity and specificity.
DNA Analysis: Circulating tumor DNA analysis has emerged as a powerful tool for cancer detection, prognosis, and treatment monitoring. By detecting tumor-specific mutations and genomic alterations in the bloodstream, ctDNA analysis provides valuable information on tumor burden, minimal residual disease, and the emergence of treatment-resistant clones.
CTC Enumeration and Characterization: Circulating tumor cells represent a rare population of cancer cells shed into the bloodstream from primary tumors or metastatic sites. By enumerating and characterizing CTCs, researchers can gain insights into tumor heterogeneity, metastatic potential, and therapeutic response. Advanced technologies such as microfluidic devices and immunomagnetic separation techniques have facilitated the isolation and analysis of CTCs with high sensitivity and specificity.
Clinical Utility and Challenges:

While blood tests for cancer detection hold immense promise, several challenges must be addressed to realize their full clinical utility. These challenges include:
Sensitivity and Specificity: Achieving optimal sensitivity and specificity in detecting circulating tumor biomarkers remains a major challenge due to the low abundance of tumor-derived molecules in the bloodstream and the presence of non-specific background signals. Improvements in assay sensitivity, specificity, and standardization are needed to enhance the clinical accuracy of liquid biopsy tests.
Biomarker Validation: The identification and validation of robust circulating tumor biomarkers are critical for the clinical implementation of blood tests for cancer detection. Biomarker discovery efforts must focus on identifying reliable markers that exhibit high sensitivity and specificity across diverse cancer types and disease stages.
Cost-Effectiveness: The cost-effectiveness of blood tests for cancer detection is an important consideration, particularly in resource-limited settings. While the upfront costs of liquid biopsy tests may be higher than traditional diagnostic modalities, their potential to reduce healthcare expenditures by enabling early detection and personalized treatment strategies warrants further evaluation.
Regulatory Approval and Reimbursement: Regulatory approval and reimbursement policies play a crucial role in the adoption of blood tests for cancer detection in clinical practice. To facilitate widespread implementation, liquid biopsy assays must undergo rigorous evaluation and receive regulatory approval from health authorities such as the FDA. Additionally, reimbursement policies must ensure adequate coverage and reimbursement for these tests to incentivize their adoption by healthcare providers.
Conclusion:
Blood tests for cancer detection represent a transformative approach to early diagnosis, personalized treatment, and disease monitoring in oncology. By leveraging the molecular information present in the bloodstream, liquid biopsies offer a non-invasive, convenient, and informative method for detecting cancer and guiding therapeutic decision-making. Despite existing challenges, ongoing advancements in technology, biomarker discovery, and regulatory oversight hold promise for the continued evolution and clinical integration of blood tests for cancer detection, ultimately improving patient outcomes and advancing the field of precision oncology.